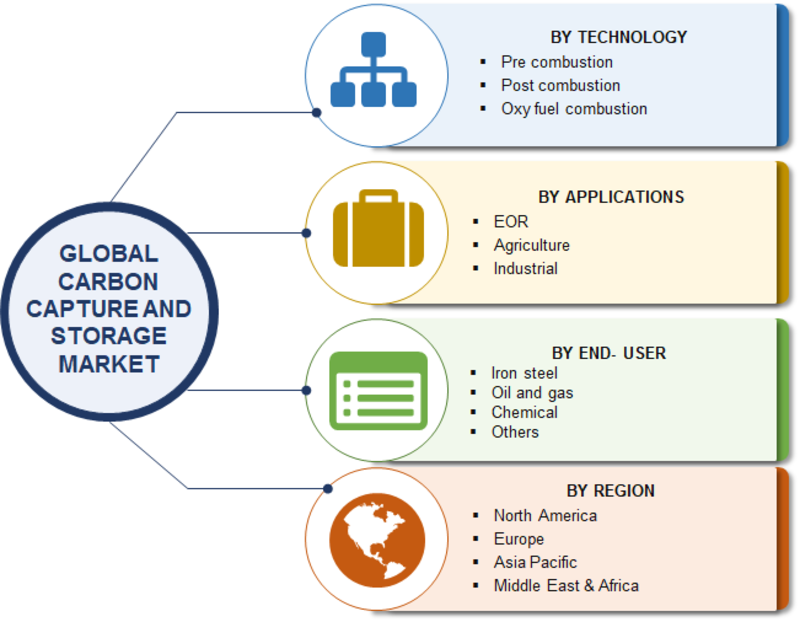
A 550-MW coal-fired power plant capturing 90% of the emitted CO 2 needs to separate about 5 Mton of CO 2 per year. CO 2 concentration in ambient air is about 0.042 percent. Iron and steel, and coal power plants emit about 20–30% and 12–15% CO 2, respectively. 
For example, natural gas and ethanol refineries emit up to 80% CO 2. Figure 1 depicts the global contributions of large industrial point sources, showing different CO 2 concentrations in the emitted gas streams. 3, 4īecause carbon capture is a multiscale problem, it is most appropriate to use different CO 2 capture methods for different emission sources. Direct air capture (DAC) of CO 2 has to grow from a few thousand tons per year to nearly 1B tons to reach net-zero emission by 2050. Based on the International Energy Agency’s (IEA) scenario, CO 2 capture from point sources has to scale up from 40M tons per year today to 6.6B tons in 2050. Thus, the development of effective methods of capturing carbon dioxide from various gas streams has become essential. The captured CO 2 can be permanently stored in the ground, or used for making useful products such as fuel, concrete, drinks, urea fertilizer, and so on. Along this line, we need technologies that can be deployed in a wide variety of situations at low cost and massive scale to capture CO 2 from power plants fired with coal or natural gas, flue gas from industrial plants, the tailpipes of cars and trucks, and legacy emissions from the air (direct air capture). 2Ĭonsidering the continuing increase in atmospheric CO 2, and the slow pace of decarbonization, carbon capture becomes a last-resort response for mitigation of global warming and climate change. In response to this global threat, in December 2015, talks at the Paris meeting have identified the need for immediate action aimed at reducing CO 2 emissions to limit the increase of global temperatures between 1.5☌ and 2☌. If this scenario materializes, the earth’s environment will become intolerable for humans and many other life forms on the planet. IPCC has shown that if the CO 2 emissions as a function of time will continue as business as usual, the rise in global surface temperature will reach 4☌. In 2021, climate anomalies, including severe drought, wildfires, flooding, melting of arctic ice, and stronger hurricanes were already experienced. 1Īccording to Berkeley Earth and UK Hadley Center, the global mean temperature in 2020 is estimated as 1.27☌ above the average temperature in the late nineteenth century. 1 Land and ocean have taken up globally about 56% per year of CO 2 emissions from human activities over the past six decades. As a result, fast and extensive variations in the atmosphere, ocean, cryosphere, and biosphere have occurred. Specifically, atmospheric carbon dioxide (CO 2) concentration now approaches 420 ppm, whereas methane (CH 4) reached 1866 ppb, and nitrous oxide (N 2O) reached 332 ppb. Greenhouse gas (GHG) emissions have increased dramatically since the industrial revolution (1750). Graphical abstractĪccording to the Intergovernmental Panel on Climate Change (IPCC) report in 2021, it is clear that human activities have warmed the atmosphere, ocean, and land. Also covered in this issue are machine learning-based computer algorithms developed with the goal to speed up the progress of carbon capture materials development, and to design advanced materials with high CO 2 capacity, improved capture and release kinetics, and improved cyclic durability. Other materials that can capture CO 2 from low concentrations of gas streams, such as air (direct air capture) are also discussed. In this issue of MRS Bulletin, some of these materials are highlighted, including solvents and sorbents, membranes, ionic liquids, and hydrides. The next wave of materials for carbon capture under investigation includes activated carbon, metal–organic frameworks, zeolites, carbon nanotubes, and ionic liquids. Another class of carbon capture materials is solid sorbents that are usually functionalized with amines or have natural affinities for CO 2. Potassium hydroxide is a benchmark aqueous sorbent that reacts with CO 2 to convert it into K 2CO 3 and subsequently precipitated as CaCO 3. Decarbonization of these sources requires special types of materials that have high affinities for CO 2. 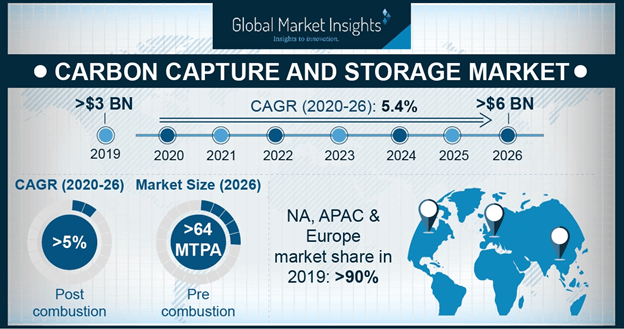
In order to combat climate change, carbon dioxide (CO 2) emissions from industry, transportation, buildings, and other sources need to be captured and long-term stored.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
